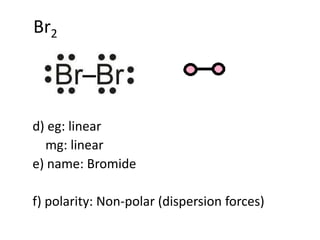
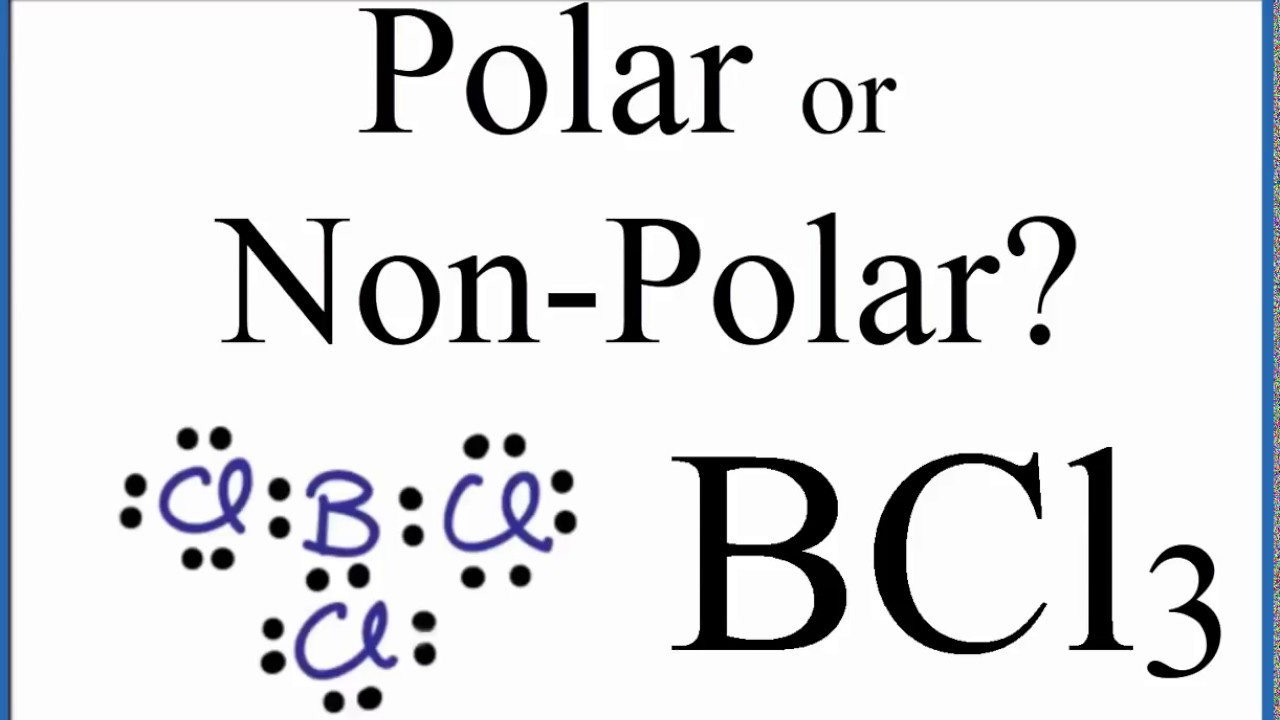Polarization of an ionic bond means the distortion of the electron cloud of an anion towards a cation Polarization of an ionic bond results in an ionic. - ppt download

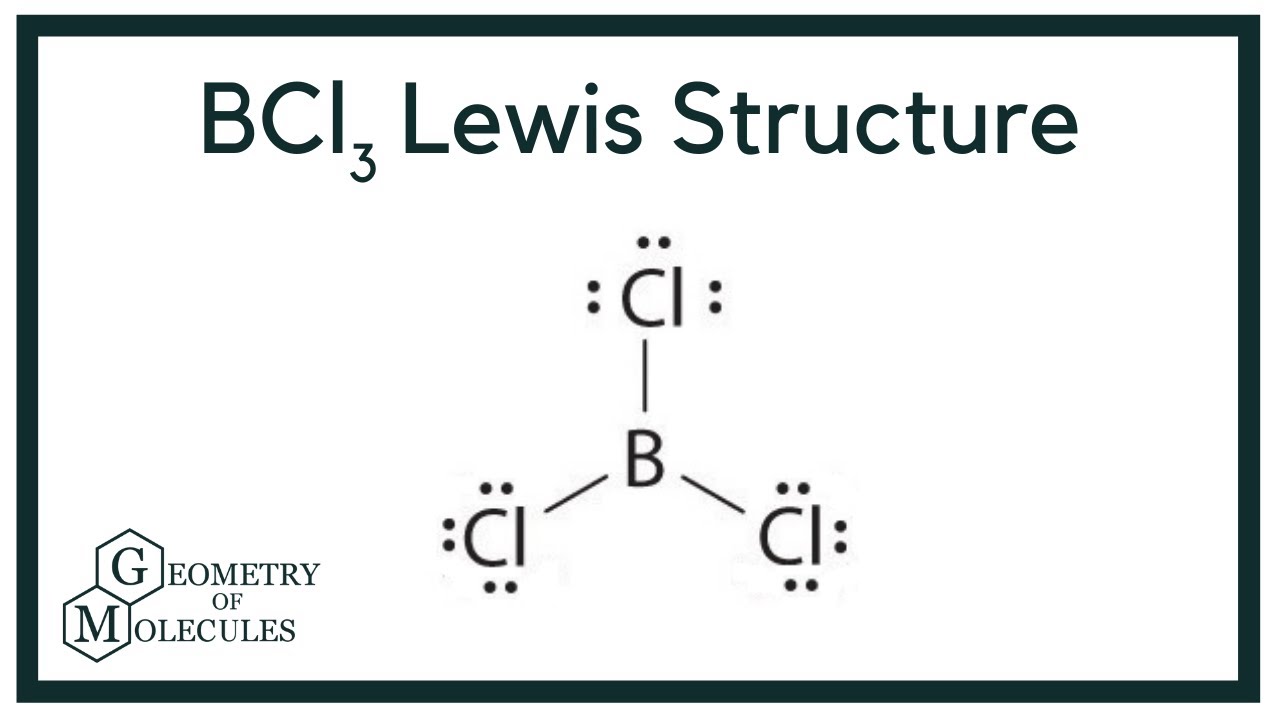
What do you expect for the B-Cl bond length in boron trichloride, BCl_3, on the basis of covalent radii? | Homework.Study.com

Consider the following statements about BCl3 molecules. Which statement is false? A. The B-Cl bonds are quite polar. B. The bond dipoles exactly cancel. C. The Cl-B-Cl bond angles are 109.5 degrees.

BCl3 lewis structure, molecular geometry, polar or nonpolar, hybridization, Bond angle | Molecular geometry, Molecular, Vsepr theory